Is C2H6O polar or nonpolar? - Polarity of C2H6O
The question is –
Is C2H6O polar or non-polar?
Answer:
⇒ C2H6O has two isomers; ethanol (C2H5OH) and dimethyl ether (CH3OCH3). Both isomers are polar molecules.
Explanation:
The polarity of C2H6O depends on the molecular arrangement of the bonded atoms (two carbon atoms, two hydrogen atoms, and one oxygen atom) and the electronegativity difference between them.
The electronegativity of each C-atom, H-atom, and O-atom is 2.55, 2.20, and 3.44, respectively.
Ethanol (C2H5OH) has a bent or V-shape, and molecular geometry as per the O-H bonded oxygen atom.
An electronegativity difference of 0.35 units exists between the bonded atoms in each C-H bond, 0.89 units in the C-O bond, and of 1.24 units exists in the O-H bond.
The oxygen atom not only attracts the shared electron cloud of the C-O and O-H bonds but also attracts electrons from each C-H bond.
As result, the oxygen atom (Oδ–) gains a partial negative charge while C-atoms (Cδ+) and H-atoms (Hδ++) attain partial positive charges.
The strong C-O and O-H dipole moments stay uncancelled in the overall molecular shape. Hence, an ethanol (C2H5OH) molecule is polar and has a high net dipole moment value (net µ > 0).
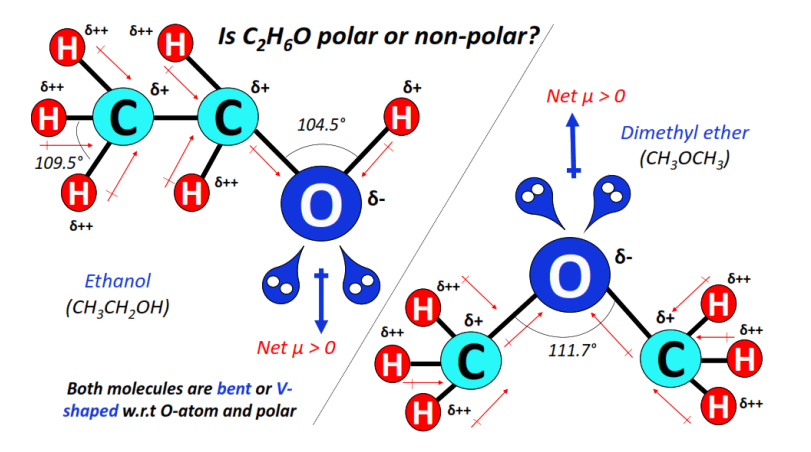
The other isomer of C2H6O i.e., dimethyl ether (CH3OCH3), also possesses a bent or V-shape w.r.t the central O-atom.
2 lone pairs of electrons on the central O-atom distort the shape and geometry of the molecule.
The dipole moments of individually polar (C-O) bonds stay uncancelled in the asymmetric, bent shape.
Hence, dimethyl ether (CH3OCH3) is also polar with a net dipole moment (net µ = 1.30 Debye).
Among the two isomers, C2H5OH is more polar than CH3OCH3 owing to the presence of a strongly polar hydroxyl (OH) functional group in it.
Also, check
- Ethanol (C2H5OH) lewis structure, molecular geometry, bond angle, hybridization
- Dimethyl ether (CH3OCH3) lewis structure, molecular geometry, bond angle, hybridization
- Is Dimethyl ether (CH3OCH3) polar or nonpolar?
⇒ How to know if a compound is polar or nonpolar?
Related Posts
- Is CH3CH2CH2OH polar or nonpolar? – Chemistry QnA
- Is OH2 polar or nonpolar? – Chemistry QnA
- Is ICl4- polar or nonpolar? – Chemistry QnA
- Is OH- polar or nonpolar? – Chemistry QnA
- Is SiS2 polar or nonpolar? – Chemistry QnA
- Is MgCl2 polar or nonpolar? – Chemistry QnA
- Is food coloring polar or nonpolar? – Chemistry QnA
- Is CaCl2 polar or nonpolar? – Chemistry QnA
- Is 2-propanol polar or nonpolar? – Chemistry QnA
- Are double bonds more polar than single bonds? – Chemistry QnA
- Is Octane (C8H18) polar or nonpolar? – Chemistry QnA
- Is S8 polar or nonpolar? – Chemistry QnA
- Is HCO3- polar or nonpolar? – Chemistry QnA
- Is SbF5 polar or nonpolar? – Chemistry QnA
- Is CH3CH2CH3 polar or nonpolar? – Chemistry QnA
- Is NH4Br polar or nonpolar? – Chemistry QnA
- Is HBrO polar or nonpolar? – Chemistry QnA
- Is SeCl2 polar or nonpolar? – Chemistry QnA
- Is IOF5 polar or nonpolar? – Chemistry QnA
- Is GeH4 polar or nonpolar? – Chemistry QnA
- Is CH4O polar or nonpolar? – Chemistry QnA
- Is KBr polar or nonpolar? – Chemistry QnA
- Is BrF polar or nonpolar? – Chemistry QnA
- Is C2H2Br2 polar or nonpolar? – Chemistry QnA
- Is SCl6 polar or nonpolar? – Chemistry QnA
- Is NO+ polar or nonpolar? – Chemistry QnA
- Is SeH2 polar or nonpolar? – Chemistry QnA
- Is Cl2O polar or nonpolar? – Chemistry QnA
- Is GaH3 polar or nonpolar? – Chemistry QnA
- Is KCl polar or nonpolar? – Chemistry QnA
- Is AlBr3 polar or nonpolar? – Chemistry QnA
- Is NaOH polar or nonpolar? – Chemistry QnA
- Is CH3CH3 polar or nonpolar? – Chemistry QnA
- Is Cyclohexane polar or nonpolar? – Chemistry QnA
- Is NH4NO3 polar or nonpolar? – Chemistry QnA
- Is SCl4F2 polar or nonpolar? – Chemistry QnA
- Is HClO polar or nonpolar? – Chemistry QnA
- Is AsH3 polar or nonpolar? – Chemistry QnA
- Cl2XeF2 polar or nonpolar? – Chemistry QnA
- SeCl6 polar or nonpolar? – Chemistry QnA
About the author
Vishal Goyal is the founder of Topblogtenz, a comprehensive resource for students seeking guidance and support in their chemistry studies. He holds a degree in B.Tech (Chemical Engineering) and has four years of experience as a chemistry tutor. The team at Topblogtenz includes experts like experienced researchers, professors, and educators, with the goal of making complex subjects like chemistry accessible and understandable for all. A passion for sharing knowledge and a love for chemistry and science drives the team behind the website. Let's connect through LinkedIn: https://www.linkedin.com/in/vishal-goyal-2926a122b/
