Is Cl2XeF2 polar or nonpolar? - Polarity of XeCl2F2
The question is –
Is Cl2XeF2 polar or non-polar?
Answer:
⇒ Cis- Cl2XeF2 is polar while trans-Cl2XeF2 is a non-polar molecule.
Explanation:
The polarity of Cl2XeF2 or XeCl2F2 depends on the arrangement of two chlorine (Cl) and two fluorine (F) atoms around the central xenon (Xe) atom.
Cis-trans isomerism exists in this molecule. In the cis-form, two Cl and two F-atoms are each present on the same side of the molecule.
Contrarily, in trans-Cl2XeF2, two Cl-atoms and two F-atoms each occupy the opposite sides of the molecule, a complementary pair facing each other.
Each Xe-Cl and Xe-F bond is individually polar owing to an electronegativity difference of 0.56 units and 1.38 units, respectively, between the bonded atoms.
Each of the Cl and F-atoms thus gains partial negative charges (δ–) while the central Xe-atom attains a partial positive charge (δ+) in the Cl2XeF2 molecule.
The Cl2XeF2 molecule possesses a square planar geometry. 2 lone pairs of electrons surround the central Xe-atom, one on each side of the molecule, which leads to an overall symmetrical shape.
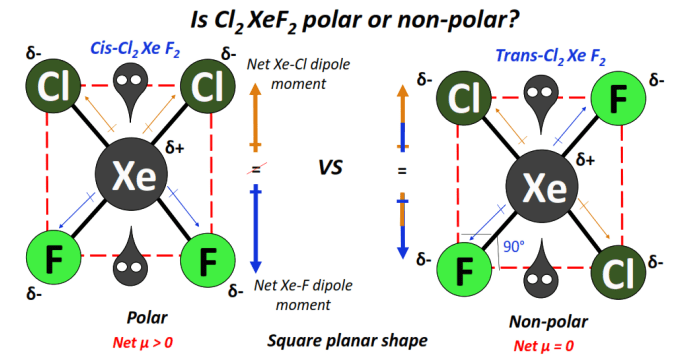
The individual Xe-Cl and Xe-F dipole moments get canceled in opposite directions in the trans-form. Thus trans-Cl2XeF2 is overall non-polar (net µ = 0).
In contrast, the stronger Xe-F dipole moment does not get canceled with the oppositely-facing Xe-Cl dipole moment in the cis-form of Cl2XeF2.
The charged electron cloud stays non-uniformly distributed. Consequently, cis-Cl2XeF2 is overall polar (net µ > 0).
⇒ How to know if a compound is polar or nonpolar?
FAQ
Is Cl2XeF2 or XeCl2F2 polar or nonpolar? |
The overall polarity of Cl2XeF2 depends on the arrangement of its atoms. The trans-form of the Cl2XeF2 or XeCl2F2 is non-polar (net µ = 0) due to the cancellation of the individual Xe-Cl and Xe-F dipole moments in opposite directions. In contrast, the cis-form of Cl2XeF2 or XeCl2F2 is polar (net µ > 0) due to the stronger Xe-F dipole moment not getting canceled with the oppositely-facing Xe-Cl dipole moment. |
What is the geometry of Cl2XeF2? |
Cl2XeF2 has a square planar geometry, meaning that the molecule is shaped like a square and all the atoms lie in the same plane. The central Xe-atom is surrounded by two lone pairs of electrons, one on each side of the molecule, which contributes to its symmetrical shape. |
What causes the polarity of Cl2XeF2? |
The polarity of Cl2XeF2 is caused by the electronegativity differences between the bonded atoms (Xe, Cl, and F). Each Xe-Cl and Xe-F bond is individually polar, with the Cl and F-atoms gaining partial negative charges (δ–) and the central Xe-atom attaining a partial positive charge (δ+). This uneven distribution of charge creates a dipole moment, which results in the overall polarity of the molecule. |
What is the difference between cis- and trans-Cl2XeF2? |
The difference between cis- and trans-Cl2XeF2 lies in the arrangement of the Cl and F-atoms around the central Xe-atom. In the cis-form, two Cl and two F-atoms are each present on the same side of the molecule. In contrast, in trans-Cl2XeF2, two Cl-atoms and two F-atoms each occupy opposite sides of the molecule, facing each other in a complementary pair. This difference in arrangement determines the net polarity of the molecule, with cis-Cl2XeF2 being polar and trans-Cl2XeF2 being non-polar. |
Related Posts
- Is CH3CH2CH2OH polar or nonpolar? – Chemistry QnA
- Is OH2 polar or nonpolar? – Chemistry QnA
- Is ICl4- polar or nonpolar? – Chemistry QnA
- Is OH- polar or nonpolar? – Chemistry QnA
- Is SiS2 polar or nonpolar? – Chemistry QnA
- Is MgCl2 polar or nonpolar? – Chemistry QnA
- Is food coloring polar or nonpolar? – Chemistry QnA
- Is CaCl2 polar or nonpolar? – Chemistry QnA
- Is 2-propanol polar or nonpolar? – Chemistry QnA
- Are double bonds more polar than single bonds? – Chemistry QnA
- Is Octane (C8H18) polar or nonpolar? – Chemistry QnA
- Is S8 polar or nonpolar? – Chemistry QnA
- Is HCO3- polar or nonpolar? – Chemistry QnA
- Is SbF5 polar or nonpolar? – Chemistry QnA
- Is CH3CH2CH3 polar or nonpolar? – Chemistry QnA
- Is NH4Br polar or nonpolar? – Chemistry QnA
- Is HBrO polar or nonpolar? – Chemistry QnA
- Is SeCl2 polar or nonpolar? – Chemistry QnA
- Is IOF5 polar or nonpolar? – Chemistry QnA
- Is GeH4 polar or nonpolar? – Chemistry QnA
- Is CH4O polar or nonpolar? – Chemistry QnA
- Is KBr polar or nonpolar? – Chemistry QnA
- Is BrF polar or nonpolar? – Chemistry QnA
- Is C2H2Br2 polar or nonpolar? – Chemistry QnA
- Is SCl6 polar or nonpolar? – Chemistry QnA
- Is NO+ polar or nonpolar? – Chemistry QnA
- Is SeH2 polar or nonpolar? – Chemistry QnA
- Is Cl2O polar or nonpolar? – Chemistry QnA
- Is GaH3 polar or nonpolar? – Chemistry QnA
- Is KCl polar or nonpolar? – Chemistry QnA
- Is AlBr3 polar or nonpolar? – Chemistry QnA
- Is NaOH polar or nonpolar? – Chemistry QnA
- Is CH3CH3 polar or nonpolar? – Chemistry QnA
- Is Cyclohexane polar or nonpolar? – Chemistry QnA
- Is NH4NO3 polar or nonpolar? – Chemistry QnA
- Is SCl4F2 polar or nonpolar? – Chemistry QnA
- Is C2H6O polar or nonpolar? – Chemistry QnA
- Is HClO polar or nonpolar? – Chemistry QnA
- Is AsH3 polar or nonpolar? – Chemistry QnA
- SeCl6 polar or nonpolar? – Chemistry QnA
About the author
Vishal Goyal is the founder of Topblogtenz, a comprehensive resource for students seeking guidance and support in their chemistry studies. He holds a degree in B.Tech (Chemical Engineering) and has four years of experience as a chemistry tutor. The team at Topblogtenz includes experts like experienced researchers, professors, and educators, with the goal of making complex subjects like chemistry accessible and understandable for all. A passion for sharing knowledge and a love for chemistry and science drives the team behind the website. Let's connect through LinkedIn: https://www.linkedin.com/in/vishal-goyal-2926a122b/
