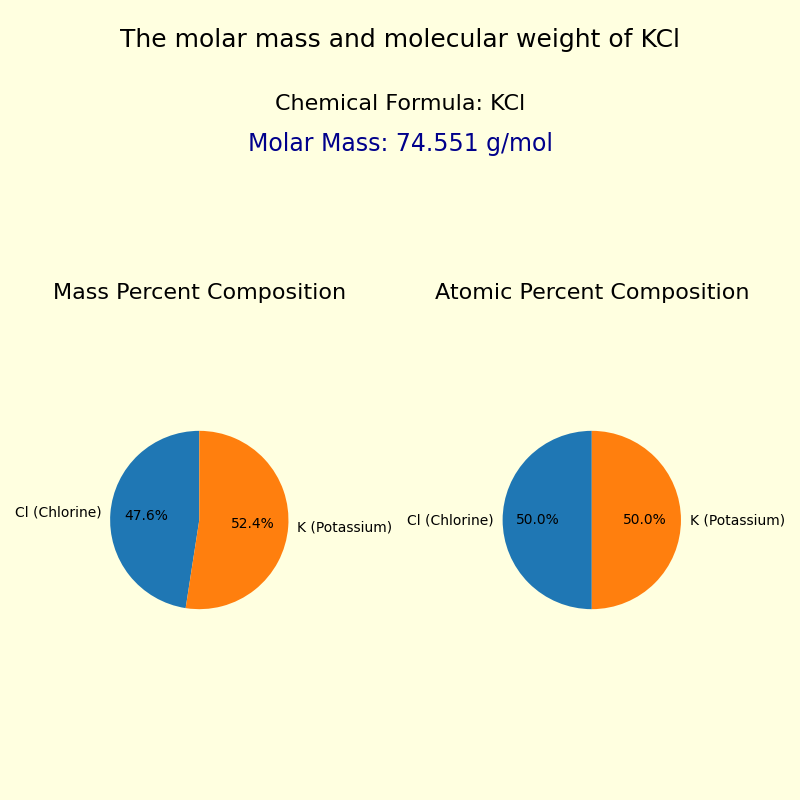
The molar mass and molecular weight of Potassium chloride (KCl) is 74.5513 g/mol.
The composition of the KCl formula is as follows:
| Element | Symbol | Atomic Weight | Atoms | Total Atomic Weight | Mass Percent |
|---|---|---|---|---|---|
| Chlorine | Cl | 35.453 g/mol | 1 | 35.4530 g/mol | 47.5552% |
| Potassium | K | 39.0983 g/mol | 1 | 39.0983 g/mol | 52.4448% |
How to find the molar mass of Potassium chloride (KCl)?
The molar mass and molecular weight of Potassium chloride (KCl) can be calculated in 4 steps.
Step I: Identify the different elemental atoms present in the KCl compound.
The given compound is KCl. It comprises atoms from 2 different elements i.e., Chlorine (Cl) and Potassium (K).
Step II: Find the atomic weight of each element in the KCl.
Here is the list of atomic weights for all elements, let’s check the Chlorine (Cl) and Potassium (K) atom’s atomic weight.
• The atomic weight of Chlorine (Cl) is 35.453 g/mol.
• The atomic weight of Potassium (K) is 39.0983 g/mol.
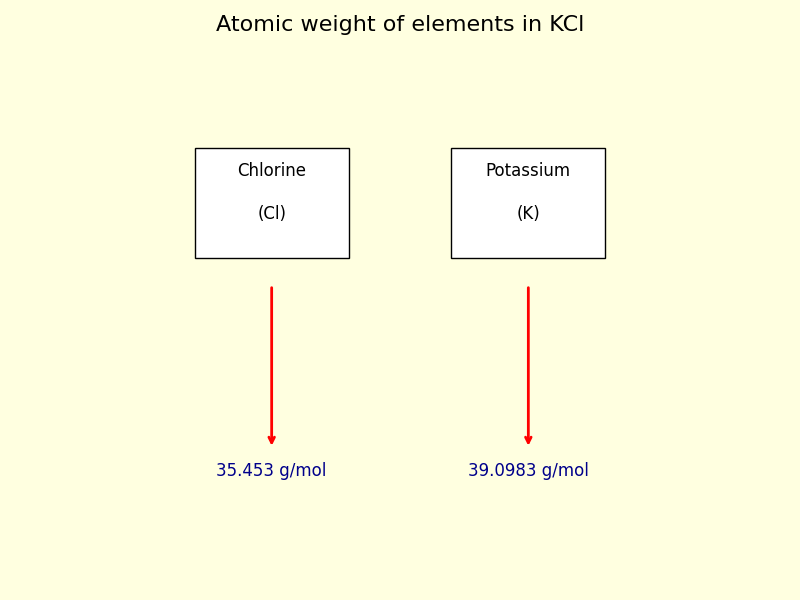
Step III: Determine the number of atoms of each element present in the KCl compound.
As per the chemical formula, KCl, It is made up of 1 Chlorine atom and 1 Potassium atom.
| Element | Number of Atoms |
|---|---|
| Cl (Chlorine) | 1 |
| K (Potassium) | 1 |
Step IV: Calculate the molar mass of the Potassium chloride (KCl) compound by applying the formula:

For Potassium chloride (KCl):
Substituting into the above formula, the values determined in steps II and III:
Molar mass of KCl = [(35.453 x 1) + (39.0983 x 1)]
Molar mass of KCl = 35.453 + 39.098 = 74.5513 g/mol
Result: The molecular weight and molar mass of Potassium chloride (KCl) is 74.5513 g/mol.
FAQs
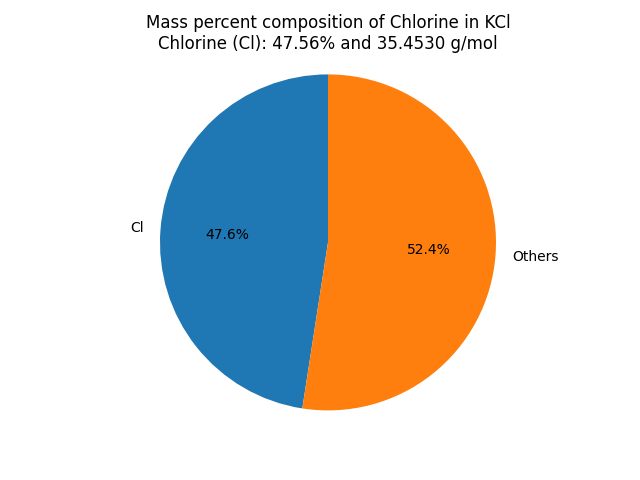
What is the mass percent composition of Chlorine (Cl) in Potassium chloride (KCl)? |
To find the mass percent of Chlorine in KCl, follow the steps given below:
∴ Mass of Chlorine in KCl = 35.453 x 1 = 35.4530 g/mol. Mass Percent Composition (%) Formula = (Mass of Element in the Compound/Molar Mass of the Compound) x 100 ∴ Mass percent of Chlorine in KCl = (Mass of Chlorine in KCl/Molar mass of KCl) × 100% Result: KCl contains 47.56 % of Chlorine as per its chemical composition.
|
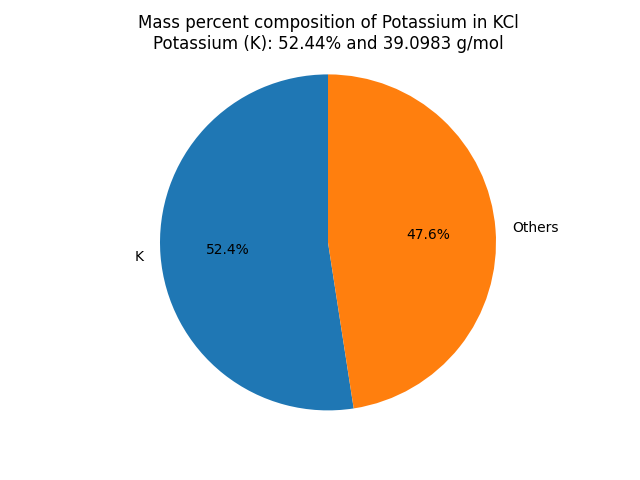
What is the mass percent composition of Potassium (K) in Potassium chloride (KCl)? |
To find the mass percent of Potassium in KCl, follow the steps given below:
∴ Mass of Potassium in KCl = 39.0983 x 1 = 39.0983 g/mol. Mass Percent Composition (%) Formula = (Mass of Element in the Compound/Molar Mass of the Compound) x 100 ∴ Mass percent of Potassium in KCl = (Mass of Potassium in KCl/Molar mass of KCl) × 100% Result: KCl contains 52.44 % of Potassium as per its chemical composition.
|
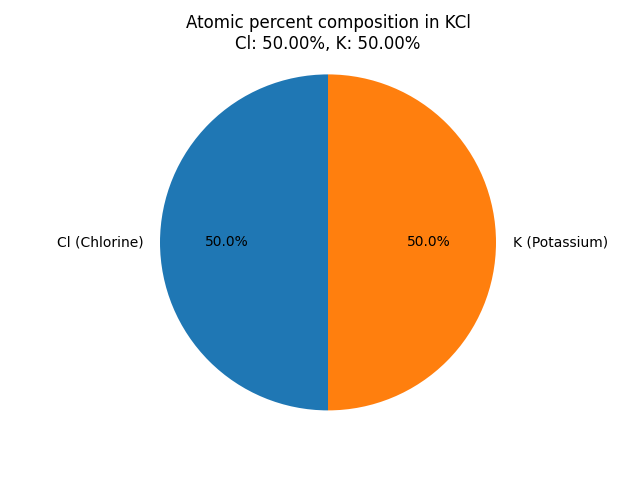
What is the atomic percentage composition of elements in Potassium chloride (KCl)? |
| Atomic percentage composition formula (%) = Number of atoms of a particular element in a compound/Total number of atoms in that compound
For KCl: The atomic percentage composition of elements in the compound is: Atomic percentage of Chlorine (Cl) in KCl: Atomic percentage of Potassium (K) in KCl:
|
How many grams of Chlorine are present in 1 mole of KCl? |
| There are 1 Cl-atoms in a KCl molecule. • Atomic weight of a Cl-atom = 35.453 g/mol ∴ Total mass of Chlorine in 1 mole of KCl = 1 x 35.453 = 35.45 g. Therefore there are 35.45 grams of Chlorine in 1 mole of KCl. |
What is the mass of 2 mol of KCl? |
| The molecular mass of KCl is 74.5513 g/mol. This means 74.5513 grams of KCl are present per mole. Therefore, we can find the mass of KCl in 2 moles as follows: ∴ Moles = Mass/Molar Mass ∴ Mass of KCl = Moles x Molar mass ∴ Mass of KCl = 2 x 74.5513 = 149.10 g Thus, the mass of 2 mol of KCl is 149.10 g. |
If you have 50.3633 grams of KCl, how many moles do you have? |
| ∴ Moles = Mass/Molar mass ∴ Moles of KCl = 50.3633/74.5513 = 0.6756 Thus, there are 0.6756 moles of KCl in its 50.3633 grams. |
What is the mass in kg of 12.43 x 1025 molecules of KCl? |
| 1 mole of a substance contains Avogadro number of particles i.e., 6.02 x 1023. Therefore, the number of moles in 12.43 x 1025 molecules of KCl are: ∴ Moles of KCl = 12.43 x 1025/6.02 x 1023 = 206.4
Now that we have its number of moles, so, we can use the molar mass of KCl (74.5513 g/mol) to find its mass as shown below. ∴ Mass of KCl = Moles x Molar mass
∴ Mass of KCl = 206.4 x 74.5513 = 15387.31 g
Converting mass from grams (g) to kilograms (kg) gives us: ∴ Mass of KCl = 15387.31/1000 = 15.39 kg
The mass of 12.43 x 1025 molecules of KCl is 15.39 kg.
|
What is the molar mass and molecular weight of Potassium chloride (KCl)? |
KCl is composed of 1 Chlorine (Cl), and 1 Potassium (K) atom.
To find the molecular mass of KCl, one multiplies the atomic weight of each element by its number of atoms in the molecule and then sums the results. ∴ For KCl, it’s (1 x 35.4530) + (1 x 39.0983).
Therefore, the molar mass of Potassium chloride (KCl) is 74.5513 g/mol.
|
About the author
Vishal Goyal is the founder of Topblogtenz, a comprehensive resource for students seeking guidance and support in their chemistry studies. He holds a degree in B.Tech (Chemical Engineering) and has four years of experience as a chemistry tutor. The team at Topblogtenz includes experts like experienced researchers, professors, and educators, with the goal of making complex subjects like chemistry accessible and understandable for all. A passion for sharing knowledge and a love for chemistry and science drives the team behind the website. Let's connect through LinkedIn: https://www.linkedin.com/in/vishal-goyal-2926a122b/